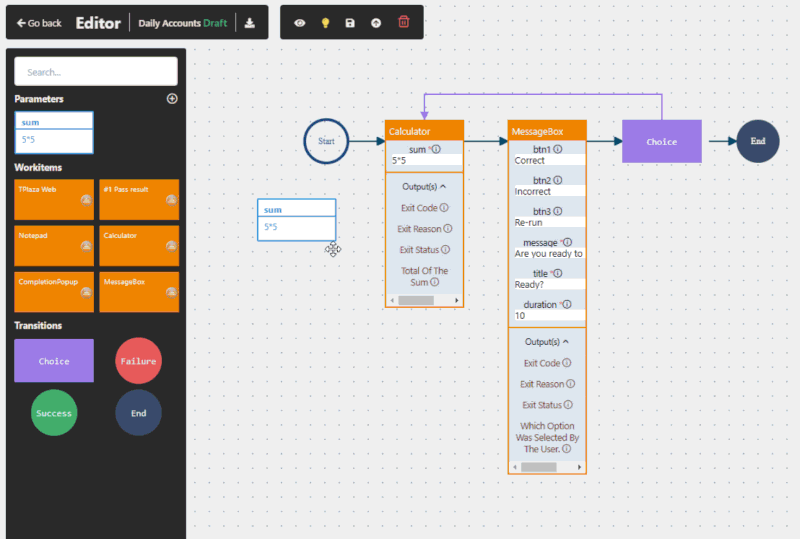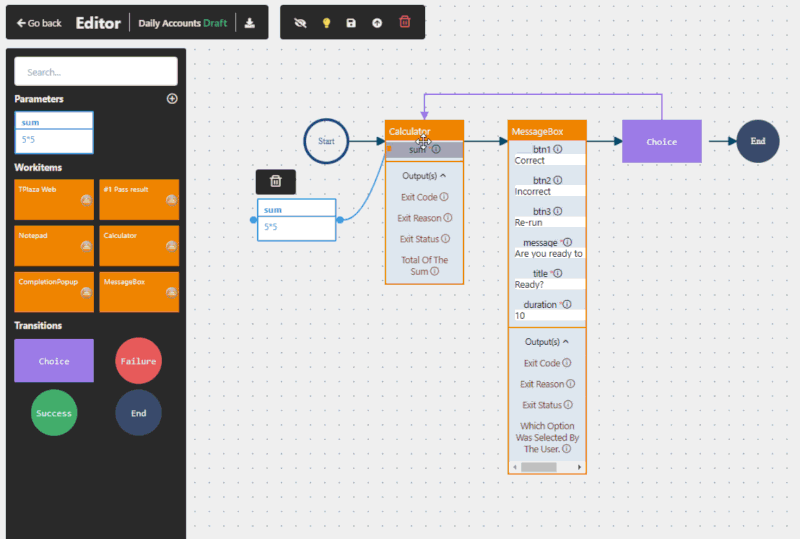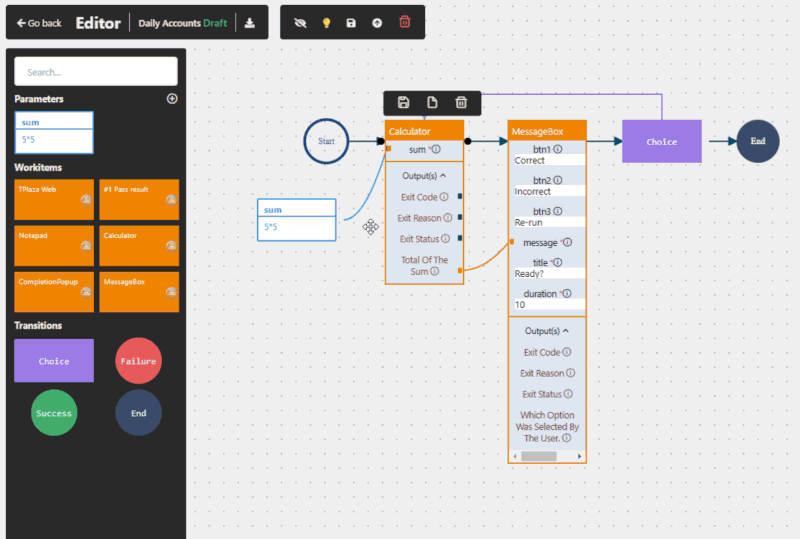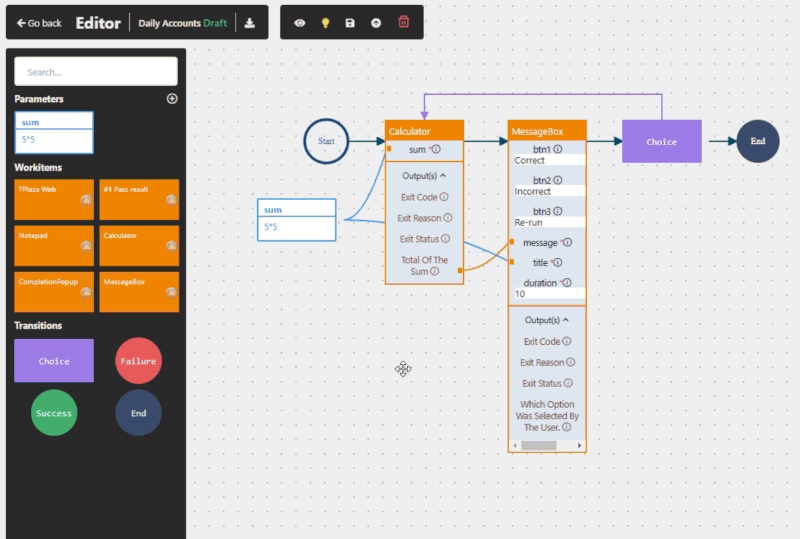
Yes, when implemented correctly. RPA in pharma operates at the user interface level, meaning it does not interfere with validated systems. This makes it suitable for regulated environments and supports compliance with frameworks such as GxP and FDA 21 CFR Part 11.
Robotic Process Automation (RPA) in Pharmaceuticles
RPA in pharma is helping organisations automate processes, improve compliance and reduce manual workloads. In the pharmaceutical industry, RPA supports accuracy, consistency and efficiency while enabling pharma regulatory RPA requirements.
- Automate compliance-driven processes with RPA in pharma to ensure accuracy and audit readiness
- Streamline operations across the pharmaceutical industry by reducing manual, repetitive tasks
- Improve efficiency and data accuracy with scalable RPA solutions in regulated environments
Built for Compliance and Reliability in Pharma
Pharmaceutical environments demand secure, reliable automation that supports strict regulatory compliance and operational efficiency. RPA in pharma reduces repetitive workloads, improves data accuracy and strengthens control across critical systems. The result is streamlined processes and scalable automation designed for the pharmaceutical industry.
Any system
or platform
Works across legacy and modern pharmaceutical systems without disruption.
Visual automation technology
Automate workflows at the user interface level, replicating real user interactions.
No code
required
Build and run automation without complex development or system changes.
Improved accuracy,
lower costs
Reduce manual errors, increase efficiency and lower operational costs.
Why Choose T-Plan for RPA in Pharma
Pharmaceutical environments require automation that is secure, reliable and compliant. T-Plan delivers RPA in pharma that works non-invasively across systems, integrates with legacy platforms and scales within regulated environments.
- Non-invasive automation
Automate processes without changing core pharmaceutical systems or applications. - Cross-platform compatibility
Run automation across desktop, web and internal pharma systems. - Automated data processing
Capture and validate data from forms, PDFs and regulatory systems. - Secure, controlled deployment
Operate on-premise or within restricted environments where compliance is critical. - Scalable automation
Expand RPA across teams without complex infrastructure changes.
What are the Benefits of RPA in Pharma?
RPA in the pharmaceutical industry uses software robots to automate repetitive, rule-based tasks such as data entry, regulatory processing and document handling. This reduces manual effort while improving accuracy, compliance and efficiency.
- Reduced manual workload
Automate high-volume, repetitive processes across operations. - Improved data accuracy
Minimise errors in critical pharmaceutical data and reporting. - Faster processing times
Accelerate workflows such as documentation, approvals and submissions. - Stronger compliance and auditability
Ensure every step is tracked and aligned with regulatory requirements. - Scalable operations
Handle increased workloads without increasing headcount.
T-Plan combines visual automation with RPA to help pharmaceutical organisations automate processes across legacy systems, modern platforms and regulatory environments from a single solution.
Secure RPA in Pharma for Compliance and Validation
Pharmaceutical organisations must automate processes without introducing risk to validated systems, regulated data or compliance frameworks. RPA in pharma operates non-invasively at the user interface level, enabling automation without system changes.
Unlike traditional automation tools that rely on complex integrations, RPA interacts directly with applications, preserving validated environments and supporting regulatory requirements. The result is secure, auditable automation aligned with the needs of the pharmaceutical industry and supporting pharma regulatory RPA.
GxP (GMP, GCP, GLP)
Regulatory frameworks governing manufacturing, clinical trials and laboratory practices.
FDA 21 CFR Part 11
Requirements for electronic records, signatures and audit trails in regulated environments.
EMA Guidelines
European regulatory standards for pharmaceutical development, approval and monitoring.
MHRA Compliance
UK regulatory authority ensuring safety, quality and efficacy of medicines.
Data Integrity (ALCOA+)
Ensuring data is attributable, legible, contemporaneous, original and accurate.
CSV (Computer System Validation)
Ensuring systems used in pharma are validated and compliant with regulatory expectations.
ICH Guidelines
International standards supporting harmonised pharmaceutical development and quality.
ISO Standards (e.g. ISO 13485)
Quality management systems for regulated medical and pharmaceutical environments.
Automating Pharma Systems in Regulated Environments
Pharmaceutical organisations must validate systems operating under strict regulatory oversight while maintaining the integrity of controlled environments and sensitive data. Automation must support these requirements without introducing risk or altering validated systems.
T-Plan’s non-invasive RPA in pharma enables organisations to automate workflows while preserving system integrity, supporting compliance and ensuring audit-ready processes across the pharmaceutical industry.
Maintaining Validation in Regulated Systems
Pharma systems operate within validated environments where changes are tightly controlled. T-Plan’s UI-driven automation works externally, allowing automation without impacting validated applications.
Supporting Computer System Validation (CSV)
Pharmaceutical organisations rely on structured validation processes. By interacting at the user interface level,
T-Plan supports repeatable automation while maintaining validated system states.
Reducing Operational Risk in Pharma Automation
Automation must be reliable and fully traceable. T-Plan ensures controlled execution, audit trails and clear visibility across automated workflows.
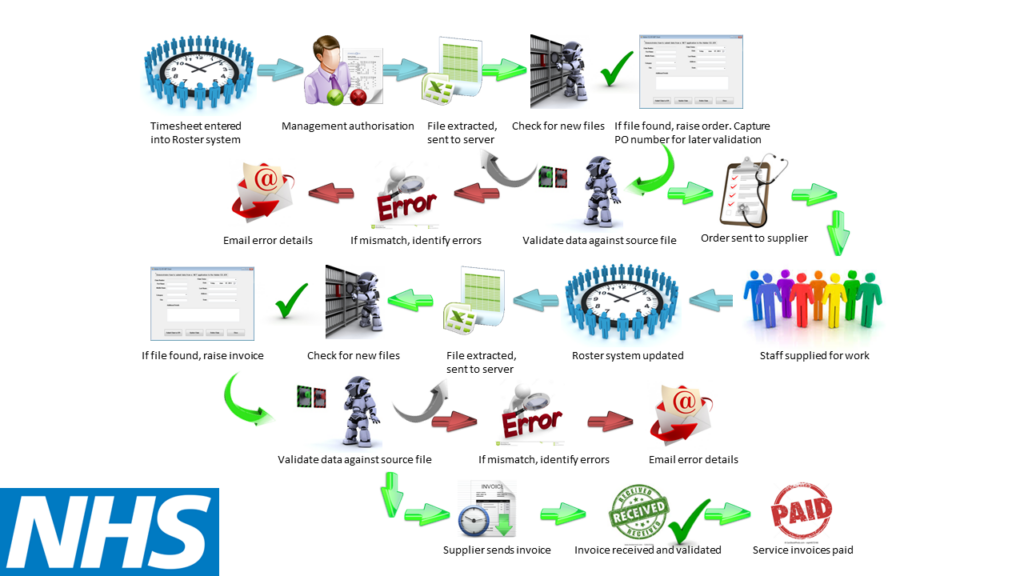
NHS Robotic Process Automation (RPA) Success Story
T-Plan was tasked with automating billing and reconciliation processes within a highly regulated healthcare environment.
Our GUI-based RPA solution reduced time spent on manual tasks and improved data accuracy by 100% within just two months.
While this example is from healthcare, the same approach to RPA in pharma enables organisations to automate complex, compliance-driven processes without impacting validated systems.

Trusted by Hundreds of Companies and Used by Thousands of People for Over 25 Years





























Key Benefits of RPA in Pharma
Non-Invasive
Automation
Automate processes through the user interface without impacting validated pharmaceutical systems.
Workflow
Automation
Streamline operational workflows across regulatory, quality and administrative functions.
Automated Data Processing
Capture and process data from forms, records and digital systems with accuracy.
Secure System
Interaction
Operate within regulated environments without requiring system changes or integrations.
Legacy System
Automation
Extend automation across legacy platforms commonly used in pharmaceutical environments.
Repeatable
Execution
Ensure consistent, reliable automation across environments and processes.
Cross-System
Automation
Coordinate workflows across multiple applications and platforms.
Audit-Ready
Tracking
Maintain clear, traceable records of all automated activity for compliance and review.
Explore Our Solutions
RPA in Pharma FAQs
RPA in pharma refers to the use of software robots to automate repetitive, rule-based processes across the pharmaceutical industry. This includes tasks such as data entry, regulatory processing and document handling, improving efficiency while maintaining compliance.
RPA in the pharmaceutical industry is used to automate workflows across areas such as clinical trials, regulatory submissions, quality assurance and administrative operations. It helps reduce manual effort, improve accuracy and accelerate processes.
By automating healthcare processes, organisations reduce manual workload while improving accuracy and processing speed.
Yes. Non-invasive RPA solutions, like those used by T-Plan, interact with applications through the user interface. This allows automation without modifying underlying systems, helping maintain validation and compliance.
Key benefits include improved data accuracy, reduced manual workloads, faster processing times, stronger compliance and the ability to scale operations without increasing headcount.
Yes. RPA can operate within secure, controlled environments and supports auditability through detailed logging and traceability of all automated actions.
RPA can be deployed quickly compared to traditional automation approaches, as it does not require complex integrations. Many organisations see value within weeks, particularly for high-volume, rule-based processes.